Ashley Guier
Idaho State University
DNP project
Chair: Dr. Jayde Barnes, DNP, APRN, PMHNP-BC, FNP-C
Committee Member: Dr. Vernon Kubiak, DNP, APRN, PMHNP-BC
Committee Member: Deb Rush, PMHNP-BC
Topic: Enhancing Early Detection and Management of Metabolic Syndrome in Patients on Second-Generation Antipsychotic Medications: A Provider-Focused Approach
Abstract
The increasing use of second-generation antipsychotics (SGAs) for mental health conditions has raised concerns due to their metabolic effects and impact on morbidity and mortality. SGAs are linked to a significantly higher risk of metabolic syndrome (MetS). Which can include central obesity, insulin resistance, dyslipidemia, hypertension, and elevated cardiovascular risk.
Among individuals with serious mental illness, MetS contributes to increased morbidity, reduced life expectancy, and challenges with treatment adherence. Despite these risks, metabolic side effects are frequently under-monitored in psychiatric settings.
Purpose: This quality improvement project aimed to evaluate psychiatric providers’ knowledge, confidence, and self-reported metabolic monitoring practices for patients prescribed SGAs through a targeted educational intervention and implementation of a standardized monitoring tool.
Methods: A single-group pretest-posttest design was implemented at an outpatient community mental health clinic in Caldwell, Idaho. Psychiatric Mental Health Practitioners (PMHNPs) participated in a 30-minute provider-focused educational session delivered through a PowerPoint presentation and guideline review. Data were collected using pre-intervention, immediate post-intervention, and one-month follow-up surveys assessing provider knowledge, confidence, perceived barriers, and self-reported monitoring practices. No patient-level data were collected.
Outcomes: Outcomes included increased provider knowledge of MetS diagnostic criteria and monitoring guidelines, improved confidence in recognizing and addressing metabolic risk, and enhanced adherence to recommended metabolic screening guidelines.
Impact: Strengthening provider awareness and consistent use of evidence-based metabolic monitoring tools may support earlier identification of metabolic abnormalities, facilitate timely referral, and reduce preventable cardiometabolic complications among patients prescribed SGAs.
Introduction
Over the past decade, the use of antipsychotic has increased substantially. Between 2013 and 2018, the number of individuals on antipsychotic drugs rose from 5 million to 6.1 million, a 22 percent increase (Ahrnsbrak & Stagnitti, 2021). SGAs are frequently preferred over FGAs because of their lower risk of extrapyramidal symptoms, which are often distressing, readily observable to others, and potentially permanent.
Despite these advantages, SGAs are strongly associated with adverse metabolic effects, including weight gain, insulin resistance, dyslipidemia, and hypertension. Collectively, these conditions contribute to MetS, which is associated with reduced life expectancy. For individuals with serious mental illness, the development of MetS can further complicate psychiatric treatment by increasing physical health burden, and negatively influencing medication adherence, and exacerbating psychological distress due to weight and chronic illness (Ospanov et al., 2019).
Metabolic changes related to SGAs often develop gradually and may go unnoticed without routine monitoring. Clinical practice guidelines from the ADA and the APA recommended baseline and ongoing metabolic monitoring for patients prescribed SGAs. The recommendations include assessment of weight, body mass index (BMI), blood pressure, hemoglobin A1C, and lipid profiles at treatment initiation and throughout treatment (American Diabetes Association et al., 2004).
Despite these guidelines, adherence to metabolic monitoring recommendation remain inconsistent in psychiatric practice. Barriers include limited appointment time, competing clinical priorities, fragmented care between psychiatric and primary care providers, uncertainty regarding provider responsibility for metabolic screening, and lack of standardized tools to support routine monitoring practices. As a result, metabolic side effects are frequently underrecognized and managed late, increasing the risk of preventable long-term complications and poorer physical health outcomes.
The purpose of this quality improvement project is to address this gap by enhancing psychiatric providers’ ability to identify metabolic syndrome in patients prescribed SGAs. This project explores whether participation in a brief educational session combined with use of the Second-Generation Antipsychotic Metabolic Monitoring tool (Appendix A) improves psychiatric providers’ knowledge, confidence, and self-reported metabolic screening practices. No patient data were reviewed, as the project focuses exclusively on provider behavior change and tool adoption.
This project explores how provider participation in a brief educational session, combined with use of the University of Washington Second-Generation Antipsychotic Metabolic Monitoring tool, can impact knowledge, awareness, and intention to monitor and refer patients with metabolic abnormalities.
Background
Bipolar disorder and schizophrenia are chronic mental health conditions that require lifelong treatment with antipsychotic medications. According to the World Health Organization, approximately 40 million people worldwide are diagnosed with bipolar disorder and, 24 million with schizophrenia, which is equivalent to 1 in 300 people globally (Mental Disorders, 2022).
Patients treated with SGAs experience substantially higher rates of MetS than antipsychotic-naïve individuals, with prevalence estimates ranging from 32% to 68% compared to 3.3% to 26%, respectively (Mortimer et al., 2023). This metabolic burden contributes to significantly reduced life expectancy in psychiatric patients, who may die seven to twenty-four years earlier than the general population (Fiorillo & Sartorius, 2021).
In response to these risks, professional organizations, including the ADA and the APA, recommend systematic metabolic monitoring for all patients receiving SGAs. Recommended baseline assessments include personal and family history of metabolic disease, measurement of weight, BMI, waist circumference, and blood pressure, laboratory evaluation of hemoglobin A1c and a fasting lipid profile before or at the time of antipsychotic initiation. Follow-up guidelines advise reassessment of BMI and waist circumference at four, eight, and twelve-weeks post-initiation, followed by quarterly BMI checks and annual or biannual laboratory work (Focht, 2025, Poojari et al., 2022).
Metabolic syndrome is diagnosed when a person has at least three of the following risk factors: abdominal obesity (waist circumference great than or equal to 40 inches for men, greater than equal inches for women, elevated triglycerides (greater than equal to 150 mg/dL, low HDL cholesterol (<40 mg/dL for men, <50 mg/dL for women), high blood pressure (greater than equal to 130/85 mm Hg), or elevated fasting glucose (greater than equal to 100 mg/dL (Dejongh, 2021). This criterion helps identify individuals at higher risk for cardiovascular disease and type 2 diabetes.
Despite these recommendations, real-world adherence to metabolic monitoring remains poor. Research indicates that many patients receiving SGAs do not obtain recommended baseline or follow-up glucose and lipid testing. Documentation of weight, BMI or waist circumference is frequently incomplete in psychiatric settings (Al-Awad et al., 2024). Barriers to adherence occur at the provider, system, and patient level and include role confusion, low confidence, lack of integrated reminders, limited laboratory access, heavy clinic workload, transportation barriers, high cost, and poor health literacy (Al-Awad et al., 2024).
SGAs differ in metabolic risk, with agents such as clozapine and olanzapine carrying greater metabolic liability compared to other SGAs. The absence of consistent monitoring may result in missed opportunities, medication adjustments, or targeted lifestyle and pharmacologic strategies. Many patients have limited primary-care contact, but maintain regular psychiatric follow-up, so psychiatric settings are uniquely positioned to serve as a focal point for metabolic screening and coordinated care (Al-Saati et al., 2025). Evaluating current adherence to metabolic monitoring guidelines and identifying setting-specific barriers and facilitators is critical to inform quality-improvement initiatives and reduce preventable cardiometabolic morbidity and mortality among patients treated with SGAs.
Literature Review
The literature review was conducted using CINAHL Complete and PubMed to identify relevant healthcare and biomedical research. A focus was placed on articles published between 2019 and 2024 to ensure inclusion of the most up-to-date evidence. Filters were applied to focus on full-text, peer-reviewed studies in English, narrowing the initial set to twenty-eight articles. The primary search terms “bipolar disorder” and “metabolic syndrome” were selected based on their relevance to the study’s objectives. Articles that focused most on the link between bipolar disorder, SGAs, and metabolic risks were included. This approach laid a solid foundation for investigating strategies to identify and treat metabolic syndrome in those taking SGAs.
Increased risk of Metabolic Syndrome
Several articles highlighted the implications of SGAs in elevating the risk of MetS among patients with mental illnesses. The metabolic side effects of SGAs include the risk for, -weight gain, insulin resistance, dyslipidemia, and hypertension, which contribute to poor long-term health outcomes and complicate the treatment of psychiatric conditions (Abo Alrob et al., 2019; Godin et al., 2024). Multiple studies demonstrated a significant increase in metabolic syndrome parameters following initiation of SGAs antipsychotics, particularly with higher-risk agents such as olanzapine and clozapine, which have been consistently associated with clinically meaningful weight gain, dyslipidemia, hypertension, and impaired glucose regulation within the first several months of treatment (Abo Alrob et al., 2019; Pillinger et al., 2020). Notably, Pillinger et al. (2020) quantified these effects, reporting that within approximately six weeks of treatment, clozapine and olanzapine were associated with a mean weight gain of approximately 3 kg and a 1 mmol/L increase in triglyceride levels, whereas metabolically lower-risk agents such as aripiprazole and ziprasidone demonstrated no significant changes compared with placebo.
While SGAs remain essential for the management of severe mental illness, their associated metabolic side effects, particularly with higher-risk agents such as olanzapine and clozapine cannot be overstated.
Barriers to Metabolic Monitoring
Despite awareness of the metabolic risks associated with SGAs, the literature reflects the challenges with adherence to metabolic monitoring protocols. Various studies highlighted the barriers to effective monitoring, such as financial constraints, unclear responsibilities among healthcare providers, and poor adherence to established guidelines (Abdulhaq et al., 2020; Al-Awad et al., 2024).
Abdulhab et al. found that although seventy-four percent of psychiatrists in Jordan were aware of metabolic monitoring guidelines, actual adherence was low, primarily due to financial limitations and patient non-compliance. Al-Awad et al.’s study in Saudi Arabia reported similar issues, with less than thirty percent of patients receiving comprehensive baseline monitoring (2024).
This low adherence reflects broader systemic issues, such as fragmented care, limited resources, and unclear roles between psychiatrists and other healthcare providers, as highlighted in Ali et al.’s (2020) systematic review. To address these issues, healthcare providers must implement coordinated plans and systemic changes to ensure comprehensive monitoring of metabolic health for patients prescribed SGAs.
Psychological and Quality of Life
Metabolic effects of SGAs not only affect physical health but can also cause psychological distress. Tandon et al. (2020) explored the real-time impact of side effects from SGAs on patients’ social, emotional, and vocational functioning. Their findings indicated that weight gain, daytime sleepiness, and sexual dysfunction led to feelings of frustration and dissatisfaction, negatively affecting patients’ self-esteem and social interactions (Tandon et al., 2020).
Similarly, Pillinger et al. (2020) noted that as psychotic symptoms improved, metabolic dysregulation worsened, suggesting a trade-off between symptom management and physical health. These research articles highlight the need to address mental and physical health outcomes, as they are closely linked to the effectiveness of SGA treatment.
Physical comorbidities, such as obesity, diabetes, stroke, and cardiovascular disease, account for approximately 60% of mortality in this population and are frequently linked to MetS (Penninx & Lange, 2018).
The economic impact of MetS among psychiatric patients extends beyond direct medical expenses. Some studies have shown that healthcare costs are as much as twenty percent more than those accrued by patients without the risk factors (Marangos et al., 2009). Patients with MetS incur higher drug expenditures, more frequent hospitalizations, and higher utilization of outpatient and physician services (Marangos et al., 2009).
SGAs impact metabolic health by interacting with dopamine, serotonin, and histamine receptors in the hypothalamus, liver, pancreas, and adipose tissue, leading to insulin resistance, fat accumulation, and impaired glucose metabolism (Carli et al., 2021). Significant weight gain associated with SGAs was first highlighted by Allison et al. in 1999, with patients gaining an average of 4.14 kg on olanzapine and 4.45 kg on clozapine within just 10 weeks. Some SGAs contribute to weight gain and others, like ziprasidone and molindone, appear to be weight neutral (Schneider et al., 2020). Patient care should be individualized and alternatives with fewer metabolic effects should be considered. Excessive weight gain remains problematic with SGA use and can contribute to psychological distress, poor patient adherence, and other health outcomes.
Genetic and lifestyle factors further shape individual risk profiles, highlighting the need for personalized treatment plans and regular metabolic monitoring (Carli et al., 2021).
To improve patient outcomes, providers must address the ongoing physical and psychological health problems using a multidisciplinary approach with dietitians, psychiatrists, primary care providers, and other specialists.
A persistent challenge in addressing metabolic risks among patients prescribed SGAs is the lack of clarity regarding provider responsibility for monitoring. Individuals with serious mental illness often have limited access to primary care but maintain regular psychiatric follow up, so there is growing recognition that psychiatric providers should assume a leading role in monitoring physical health needs, and provide appropriate referrals, when indicated.
Strategies for Improving Metabolic Health Monitoring
Several research studies explored strategies to increase adherence to metabolic monitoring standards, from improved provider education to clinical decision support technologies.
Cohen et al. (2020) demonstrated that implementing the Best Practice Alert (BPA) system in an outpatient setting improved lab ordering for metabolic monitoring from 14.4% with passive alerts to 54.3% with active alerts. The BPA system, triggered during medication order entry, has proven to be highly effective and it has the potential to bridge monitoring gaps.
In addition, professional organizations have advocated for updated training programs incorporating physical health management in psychiatric care (Ali et al., 2020; Abdulhaq et al., 2020). A combination of technology-driven solutions and provider education can help address systemic barriers and improve metabolic health outcomes for patients on SGAs.
Predictors of Metabolic Syndrome
Studies also identify specific predictors of MetS, which can inform targeted interventions. Age, gender, type, duration of SGA use, and lifestyle habits (smoking and alcohol use) can increase the risk of developing MetS (Godin et al., 2024; Amogne et al., 2021).
Godin et al.’s longitudinal study on individuals with bipolar disorder, for instance, found that males, older adults, and those with lifestyle risk factors were more likely to develop MetS over time (2024). Customizing monitoring and intervention efforts for high-risk groups could be an effective strategy to mitigate the impact of metabolic side effects.
Theoretical Model
The Stetler model examines how to use evidence-based research that can drive formal change within healthcare organizations. It provides a structured framework at both the practitioner and organizational level, supporting individual practitioners that can use research in integrating critical thinking and reflective practice (Stetler, 2001). The model consists of five phases including preparation, validation, comparative evaluation/decision making, translation/application, and evaluation (Stetler, 2001).
The Stetler model served as the framework for guiding this quality improvement project. This model is a valuable framework for improving metabolic syndrome detection and management among patients taking SGAs. The preparation phase included a review of current evidence-based practices and the identification of gaps in adherence screening protocols.
The validation phase incorporated appraisal of relevant literature, such as studies on the effectiveness of routine metabolic screening tools and provider education in improving early detection.
Evidence was compiled during the comparative evaluation and decision-making stage to ascertain the viability and relevance of incorporating a systematic metabolic monitoring protocol. This phase guided a provider-focused educational module that reviewed metabolic syndrome risk factors and early indicators.
During the translation/application phase, practitioners were trained to use standardized screening tools and implement this evidence-based approach into practice. During the evaluation phase, provider knowledge and screening rates were measured by pretest-posttest comparisons to gauge the effectiveness of the intervention. This project offered a systematic, evidence-based strategy to improve early detection and management of metabolic syndrome by integrating the Stetler Model, with the goal of improving patient outcomes.
Methodology
Implementation/Methods: This quality improvement project implemented a provider-focused educational intervention designed to increase awareness, confidence, and engagement in MetS screening among psychiatric providers who prescribe SGAs. A single-group pretest-posttest design was used to evaluate changes in provider knowledge, confidence, and self-reported metabolic monitoring practices following the intervention. No patient-level data was collected.
Setting: The project was conducted at Renewed Hope Counseling and Wellness (RHCW) in Meridian, Idaho, an outpatient community mental health clinic that provides psychiatric and counseling services for adults and children with a range of mental health conditions. Providers at this site routinely prescribe SGAs and are therefore well positioned to support early identification and referral for metabolic abnormalities.
Population: The target population consists of PMHNPs and Family Nurse Practitioners (FNPs) at RHCW who prescribe SGAs as part of their clinical care. Participation was voluntary and providers were eligible if they routinely prescribe SGAs, and no exclusion criteria were applied. To maintain participant confidentiality, individuals were instructed to refrain from including identifying information, such as their names, on the assessment instruments.
Educational Intervention: The intervention consisted of a brief, 30-minute in-person educational session delivered using a PowerPoint presentation and guided discussion. Educational materials included PowerPoint slides and a printed copy of the Second-Generation Antipsychotics Metabolic Monitoring tool (Appendix B). The educational session addressed key topics related to metabolic syndrome, including MetS diagnostic criteria, associated health risks, and the metabolic effects of SGAs. Evidence-based metabolic guidelines from the ADA and the APA were reviewed, with emphasis on recommend baseline and ongoing monitoring parameters.
Additional content included referral thresholds for metabolic abnormalities, common provider-and system-level barriers to routine screening, and strategies for integrating metabolic monitoring into psychiatric clinical workflows. Instruction and demonstration on use of the screening tool were provided to support consistent implementation in practice.
Following the session, providers were encouraged to use the screening tool during clinical encounters and to provide informal feedback regarding its clarity, relevance, and ease of use.
Data Collection: Prior to the educational intervention, participants completed a survey (Appendix C) assessing baseline knowledge of metabolic syndrome, confidence in identifying metabolic risks associated with SGAs, current metabolic monitoring practices, and perceived barriers to routine screening. Immediately following the educational session, participants completed a post-intervention survey to allow comparison of knowledge change. Additional items assessed include provider confidence, intention to change metabolic monitoring practices, and satisfaction with the education session. The post-intervention survey included three open-ended qualitative questions (Appendix D) exploring perceived usefulness of the educational content, anticipated barriers, facilitators, and suggestions for improvement.
One month after the educational intervention, participants completed a follow-up survey (Appendix E) assessing self-reported changes in metabolic monitoring practices, frequency of screening tool use, persistent barriers to implementation, and additional feedback regarding feasibility and sustainability.
Tools
This quality improvement project utilized the Second-Generation Antipsychotic Metabolic Monitoring Tool (Appendix B) to support guideline-concordant metabolic monitoring for patients prescribed SGAs and to reinforce provider awareness of key metabolic risk indicators recommended by the ADA and APA. The tool consists of a standardized checklist for monitoring metabolic parameters, including vital signs, height, weight, BMI, waist circumference, fasting glucose, and lipid profile. In this project, it was used as an educational and workflow support resource rather than a data collection instrument, and no patient charts or laboratory data were accessed.
The tool was implemented at RHCW, and adapted to available clinic resources, with emphasis on routine monitoring of blood pressure, height, weight, BMI, and waist circumference. Its use was intended to support early identification of metabolic abnormalities, facilitate timely referral, and promote more consistent application of evidence-based monitoring practices among psychiatric providers.
Quantitative survey data were analyzed using descriptive statistics. Pre- and post-intervention knowledge scores were compared by examining changes in the number and percentage of correct responses. Provider confidence, intention to change practice, perceived barriers, and satisfaction were summarized using frequency distributions of Likert-scale responses. Follow-up survey data were analyzed descriptively to assess self-reported changes in monitoring practices and use of the screening tool. Qualitative data obtained from open-ended survey questions were analyzed using basic thematic analysis. Responses were reviewed, coded, and grouped into common themes related to perceived usefulness of education intervention, barriers to implementation, facilitators of metabolic monitoring, and recommendations for improvement.
Outcomes
Six PMHNPs at RHCW completed both the pre- and post-intervention assessments, with all participants completing the one-month follow-up survey. Knowledge scores were calculated as the total number of correct responses across eight multiple-choice items (range 0-8). Pre-intervention scores ranged from 4 to 7 (M = 5.5, median = 5.5).
Following the educational session, scores increased to a range of 7 to 8 (M = 7.7, median = 8), with all participants demonstrating improvement (mean increase = 2.17).
A Wilcoxon signed-rank test was used to compare pre- and post-intervention scores. Results showed a statistically significant improvement following the intervention (W= 0, Z = -2.20, p = .031, two-tailed). The effect size was large (r = 0.90), suggesting a meaningful increase in provider knowledge despite the small sample.
Provider knowledge improved across all assessed domains following the educational intervention (Figure 1). At baseline, the lowest scores were seen in metabolic syndrome prevalence, waist circumference criteria, fasting glucose thresholds, and recommended monitoring intervals, with about 50 percent of participants answering correctly. These areas showed the greatest improvement after the intervention, with correct responses increasing to nearly 100 percent.
At one-month follow-up, knowledge scores remained high across all items (83 to 100 percent), indicating that most of the knowledge gained was maintained over time. Items with higher baseline accuracy remained stable across all time points. Participants represented a range of clinical experience levels and patient populations. Most providers reported less than one year of experience (n=3), followed by one to five years (n=2), and one provider with more than ten years of experience. All participants reported caring for adult patients (n=6), with additional overlap in adolescent (n=4) and pediatric (n=4) populations. Most providers reported prescribing SGAs between six and ten patients per week, (n=3), followed by one to five patients (n=2) and one provider reporting more than ten patients.
At baseline, metabolic monitoring practices were variable across providers. Most participants reported “often” or “sometimes” obtaining baseline metabolic measurements, monitoring weight and BMI, and assessing blood pressure. However, laboratory monitoring practices were less consistent, with most providers reporting “sometimes” reviewing or ordering fasting glucose and lipid panels.
Participants reported more consistent use of guideline-based monitoring across all domains following the intervention. Providers described increased use of baseline assessments, more regular monitoring of weight and blood pressure, and more consistent ordering of fasting glucose and lipid panels. In addition, several workflow changes were implemented at the clinic level, including the introduction of measuring tapes for waist circumference and the addition of a dedicated section in the EHR to document metabolic monitoring and last time blood work was drawn.
At baseline, commonly reported barriers included time constraints (n=2), patient nonadherence to laboratory testing (n=2), and lack of standardized tools (n=2). One participant also reported uncertainty regarding responsibility between psychiatric and primary care providers. Following the intervention, fewer participants identified lack of standardized tools as a barrier, suggesting that the implementation of the metabolic monitoring tool helped to address this gap. However, time constraints and patient adherence remained persistent challenges at both post-intervention and one-month follow-up.
At one-month follow-up, five participants achieved perfect scores, while one scored slightly lower, consistent with the overall high level of retained knowledge. Providers reported continued use of the metabolic monitoring tool and ongoing incorporation of guideline-based practices into clinical care. Qualitative responses supported these findings, with participants noting improved clarity regarding metabolic syndrome criteria and monitoring intervals. The structured monitoring tool was described as helpful in organizing workflow and supporting more consistent screening practices. Despite these improvements, participants continued to report challenges related to limited time during visits and difficulty ensuring patient follow-through with laboratory testing.
Figure 1
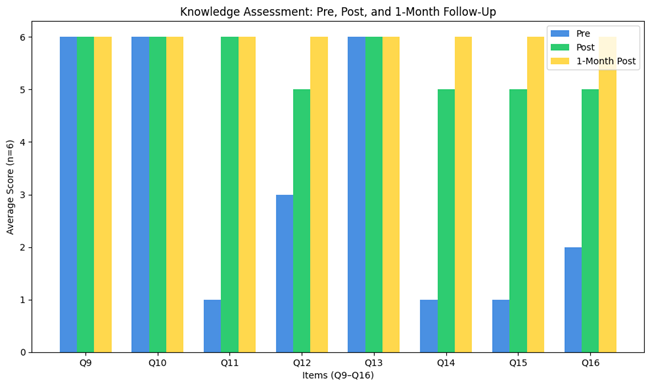
Figure 1. Changes in total provider knowledge scores from baseline to post-intervention and one-month follow-up.
Figure 2
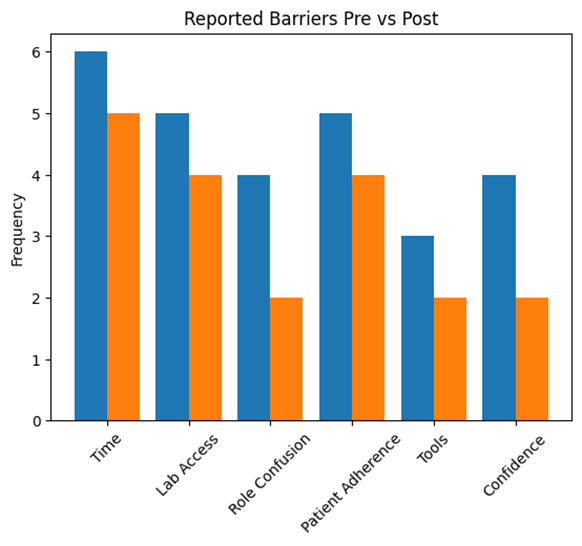
Figure 2. Reported barriers to metabolic monitoring before and after the educational intervention.
Table 1. Provider Characteristics
| Characteristic | n (%) |
| Years in Practice | |
| <1 year | 1 (12.5) |
| 1–5 years | 3 (37.5) |
| 6–10 years | 2 (25.0) |
| >10 years | 2 (25.0) |
| Patient Population Served* | |
| Adults | 8 (100) |
| Adolescents | 5 (62.5) |
| Children | 3 (37.5) |
| SGA Prescriptions per Week | |
| 1–5 | 2 (25.0) |
| 6–10 | 4 (50.0) |
| >10 | 2 (25.0) |
Table 1. Demographic and practice characteristics of participating providers (N=8).
Table 2. Practice Behaviors
| Item | Time | Never n (%) | Rarely n (%) | Sometimes n (%) | Often n (%) | Always n (%) |
| Baseline metabolic monitoring | Pre | 2 (25.0) | 2 (25.0) | 3 (37.5) | 1 (12.5) | 0 (0) |
| Post | 0 (0) | 1 (12.5) | 2 (25.0) | 3 (37.5) | 2 (25.0) | |
| Weight/BMI monitoring | Pre | 1 (12.5) | 2 (25.0) | 3 (37.5) | 2 (25.0) | 0 (0) |
| Post | 0 (0) | 0 (0) | 2 (25.0) | 4 (50.0) | 2 (25.0) | |
| Blood pressure monitoring | Pre | 1 (12.5) | 1 (12.5) | 4 (50.0) | 2 (25.0) | 0 (0) |
| Post | 0 (0) | 0 (0) | 2 (25.0) | 3 (37.5) | 3 (37.5) | |
| Glucose/lipid monitoring | Pre | 2 (25.0) | 2 (25.0) | 3 (37.5) | 1 (12.5) | 0 (0) |
| Post | 0 (0) | 1 (12.5) | 2 (25.0) | 3 (37.5) | 2 (25.0) |
| Barrier | Pre n (%) | Post n (%) |
| Time constraints | 6 (75.0) | 5 (62.5) |
| Access to laboratory testing | 5 (62.5) | 4 (50.0) |
| Role confusion | 4 (50.0) | 2 (25.0) |
| Patient nonadherence | 5 (62.5) | 4 (50.0) |
| Lack of standardized tools | 3 (37.5) | 2 (25.0) |
| Limited confidence/knowledge | 4 (50.0) | 2 (25.0) |
Table 2. Practice Behaviors and Reported Barriers Before and After the Educational Intervention.
Discussion
This quality improvement project examined whether a brief educational intervention could improve provider knowledge and awareness of recommended metabolic monitoring practices for patients prescribed SGAs. Results showed a statistically significant improvement in knowledge following the session.
Before the intervention, responses reflected some variability in familiarity with recommended monitoring practices. After the session, providers reported a better understanding of guideline-based screening and greater confidence in applying these practices during patient visits. These findings suggest that even a brief, focused educational session can help address knowledge gaps and improve comfort with metabolic monitoring. Providers also described making practical changes to their workflow, including incorporating waist circumference measurements, using a designated section in the EHR to document monitoring, and relying more directly on guideline recommendations when deciding when to order labs. These changes suggest that the intervention did more than improve knowledge and may have helped shift day-to-day practice in a meaningful way.
Feedback from participants helped provide additional context. Several noted that reviewing metabolic syndrome criteria and recommended monitoring intervals made it easier to understand what should be done and when. Others described the monitoring tool as helpful in organizing care and making screening more consistent. At the same time, ongoing barriers were still present. Time constraints during visits and difficulty with patient follow-through on lab work continued to affect consistent monitoring.
These findings are consistent with prior studies in psychiatric settings, where lack of time and challenges with lab completion are commonly reported barriers. While the educational session appeared to reduce reliance on informal or inconsistent approaches-particularly by addressing the lack of a standardized tool-it did not eliminate broader system-level challenges.
Overall, the findings of this project support the use of targeted educational interventions to improve provider awareness and confidence related to metabolic monitoring. However, education alone is unlikely to fully address barriers related to workflow and patient adherence. Continued efforts to integrate practical tools, reinforce guideline use, and address system-level factors may help improve long-term consistency in metabolic screening practices.
Limitations
This quality improvement project has several limitations that should be considered when interpreting the results. The most notable limitation is the small sample size (n=6), which limits statistical power and the precision of the findings. Although a statistically significant improvement in provider knowledge was observed, results derived from small samples must be interpreted cautiously. The effect size may appear large, but with a limited number of participants, estimates are less stable and not broadly generalizable.
The project also employed a single-group pretest-posttest design without a comparison group. While improvements in knowledge and self-reported practices followed the educational intervention, it is not possible to attribute these changes solely to the intervention. Exposure to the pretest itself may have increased awareness of metabolic monitoring concepts, contributing to improved post-test performance. Repeated exposure to the same instrument may also have influenced responses.
This project focuses on provider knowledge and perceived practice behaviors rather than objective patient outcomes. As such, it cannot determine whether improvements in knowledge translated into measurable changes in metabolic screening rates or patient health indicators such as weight, lipid levels, or glucose control. Additionally, the one-month follow-up period may not be sufficient to determine whether reported practice changes are sustained over time.
The study was conducted within a single outpatient psychiatric clinic, which may limit applicability to other settings. Differences in patient populations, available resources, staffing models, and electronic health record systems may influence the feasibility and effectiveness of similar interventions elsewhere.
Reliance on self-reported data introduces the possibility of recall bias and social desirability bias. Following an educational intervention that emphasized guideline adherence participants may have unintentionally overreported improvements in their monitoring practices. Incorporating objective measures such as chart audits or laboratory ordering data would provide a more accurate assessment of practice change. There is also the potential for self-selection bias, as providers who agreed to participate may have been more motivated or more interested in metabolic monitoring practices than those who did not participate. This may limit how well the finding reflect the broader provider population.
Finally, while the intervention addressed knowledge gaps and introduced a structured monitoring tool, it did not directly modify broader system-level barriers such as workflow demands, limited laboratory access, competing clinical priorities, or institutional policies.
These structural factors may continue to influence long-term adherence to metabolic monitoring recommendations. Future initiatives should consider larger, multisite samples, longer follow-up periods and incorporation of objective clinical outcome measures. Including a comparison group would strengthen causal inference and provide clearer evidence regarding the sustained impact of provider-focused metabolic monitoring education.
Conclusion
This quality improvement project showed that a brief educational intervention improved provider knowledge of metabolic monitoring practices for patients receiving SGAs. Following the session, participants demonstrated clear gains in knowledge and greater familiarity with guideline-based monitoring. Providers also described increased confidence in incorporating these practices into routine care. In addition to these changes, providers reported making small but meaningful adjustments to their workflow, including incorporating waist circumference measurements, using the EHR to document monitoring more consistently, and relying more directly on guideline recommendations when ordering labs.
Despite these improvements, barriers such as time constraints during visits and patient adherence with laboratory testing remained. These findings suggest that while education can improve knowledge and support changes in practice, additional workflow and system-level supports are likely needed to maintain consistent metabolic monitoring.
Overall, this project supports the use of targeted education as a practical first step in improving metabolic monitoring practices. Continued efforts to reinforce guideline use and address ongoing barriers may help promote more consistent improvements in long-term patient outcomes.
References
Abdulhaq, B., Dardas, L. A., & Sami, O. (2020). Monitoring for the metabolic side effects of second‐generation antipsychotic medications: Psychiatrists’ views and practices. Perspectives in Psychiatric Care, 57(3), 1237–1243. https://doi.org/10.1111/ppc.12679
Abo Alrob, O., Alazzam, S., Alzoubi, K., Nusair, M. B., Amawi, H., Karasneh, R., Rababa’h, A., & Nammas, M. (2019). The effect of long-term second-generation antipsychotics use on the metabolic syndrome parameters in jordanian population. Medicina, 55(7), 320. https://doi.org/10.3390/medicina55070320
Ahrnsbrak, R., & Stagnitti, M. N. (2021, February). Comparison of antidepressant and antipsychotic utilization and expenditures in the U.S. civilian noninstitutionalized population, 2013 and 2018. Agency for Healthcare Research and Quality. https://meps.ahrq.gov/data_files/publications/st534/stat534.shtml#:~:text=Between%202013%20and%202018%2C%20the,change%20was%20not%20statistically%20significant.
Al-Awad, F. A., Almutairi, H. A., Almutairi, S. A., Alessa, O. S., Alanazi, S. F., Alzain, N. M., Albakr, D. M., & Alzahrani, S. I. (2024). Adherence to the monitoring of metabolic syndrome in patients receiving antipsychotics in outpatient clinics in saudi arabia. Journal of Family and Community Medicine, 31(1), 42–47. https://doi.org/10.4103/jfcm.jfcm_153_23
Ali, R., Jalal, Z., & Paudyal, V. (2020). Barriers to monitoring and management of cardiovascular and metabolic health of patients prescribed antipsychotic drugs: A systematic review. BMC Psychiatry, 20(1). https://doi.org/10.1186/s12888-020-02990-6
Allison, D. B., Mentore, J. L., Heo, M., Chandler, L. P., Cappelleri, J. C., Infante, M. C., & Weiden, P. J. (1999). Antipsychotic-induced weight gain: A comprehensive research synthesis. American Journal of Psychiatry, 156(11), 1686–1696. https://doi.org/10.1176/ajp.156.11.1686
Amogne, G., Alemu, T., Tadesse, T., & Mullu, A. (2021). Magnitude of metabolic syndrome and its predictors among patients on second-generation antipsychotic drugs at six psychiatry clinics and mental hospitals, in addis ababa, ethiopia, 2019; multicenter cross-sectional study. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 15(4), 102187. https://doi.org/10.1016/j.dsx.2021.102187
Carli, M., Kolachalam, S., Longoni, B., Pintaudi, A., Baldini, M., Aringhieri, S., Fasciani, I., Annibale, P., Maggio, R., & Scarselli, M. (2021). Atypical antipsychotics and metabolic syndrome: From molecular mechanisms to clinical differences. Pharmaceuticals, 14(3), 238. https://doi.org/10.3390/ph14030238
Clark, N. G. (2004). Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care, 27(2), 596–601. https://doi.org/10.2337/diacare.27.2.596
Cohen, S., Bostwick, J. R., Marshall, V. D., Kruse, K., Dalack, G. W., & Patel, P. (2020). The effect of a computerized best practice alert system in an outpatient setting on metabolic monitoring in patients on second‐generation antipsychotics. Journal of Clinical Pharmacy and Therapeutics, 45(6), 1398–1404. https://doi.org/10.1111/jcpt.13236
DeJongh, B. M. (2021). Clinical pearls for the monitoring and treatment of antipsychotic induced metabolic syndrome. Mental Health Clinician, 11(6), 311–319. https://doi.org/10.9740/mhc.2021.11.311
Fiorillo, A., & Sartorius, N. (2021). Mortality gap and physical comorbidity of people with severe mental disorders: The public health scandal. Annals of General Psychiatry, 20(1). https://doi.org/10.1186/s12991-021-00374-y
Godin, O., Olié, E., Fond, G., Aouizerate, B., Aubin, V., Bellivier, F., Belzeaux, R., Courtet, P., Dubertret, C., Haffen, E., Lefrere, A., Llorca, P. M., Polosan, M., Roux, P., Samalin, L., Schwan, R., Leboyer, M., & Etain, B. (2024). Incidence and predictors of metabolic syndrome onset in individuals with bipolar disorders: A longitudinal study from the face‐bd cohort. Acta Psychiatrica Scandinavica. https://doi.org/10.1111/acps.13655
Marangos, P. J., Okamoto, L. J., & Caro, J. J. (2009). Economic burden of the components of the metabolic syndrome. In Handbook of disease burdens and quality of life measures (pp. 1135–1149). Springer New York. https://doi.org/10.1007/978-0-387-78665-0_64
Mental disorders. (2022, June 8). https://www.who.int/news-room/fact-sheets/detail/mental-disorders
Mortimer, K. H., Katshu, M., & Chakrabarti, L. (2023). Second-generation antipsychotics and metabolic syndrome: A role for mitochondria. Frontiers in Psychiatry, 14. https://doi.org/10.3389/fpsyt.2023.1257460
Ospanov, O., Yeleuov, G., Kadyrova, I., & Bekmurzinova, F. (2019). The life expectancy of patients with metabolic syndrome after weight loss: Study protocol for a randomized clinical trial (lifexpe-rt). Trials, 20(1). https://doi.org/10.1186/s13063-019-3304-9
Panagiotopoulos, C., Ronsley, R., Kuzeljevic, B., & Davidson, J. (2012). Waist circumference is a sensitive screening tool for assessment of metabolic syndrome risk in children treated with second-generation antipsychotics. The Canadian Journal of Psychiatry, 57(1), 34–44. https://doi.org/10.1177/070674371205700107
Penninx, B. H., & Lange, S. M. (2018). Metabolic syndrome in psychiatric patients: Overview, mechanisms, and implications. Dialogues in Clinical Neuroscience, 20(1), 63–73. https://doi.org/10.31887/dcns.2018.20.1/bpenninx
Pillinger, T., McCutcheon, R. A., Vano, L., Mizuno, Y., Arumuham, A., Hindley, G., Beck, K., Natesan, S., Efthimiou, O., Cipriani, A., & Howes, O. D. (2020). Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia, predictors of metabolic dysregulation, and association with psychopathology: A systematic review and network meta-analysis. The Lancet Psychiatry, 7(1), 64–77. https://doi.org/10.1016/s2215-0366(19)30416-x
Schneider, M., Pauwels, P., Toto, S., Bleich, S., Grohmann, R., Heinze, M., & Greiner, T. (2020). Severe weight gain as an adverse drug reaction of psychotropics: Data from the amsp project between 2001 and 2016. European Neuropsychopharmacology, 36, 60–71. https://doi.org/10.1016/j.euroneuro.2020.05.001
Stetler, C. B. (2001). Updating the stetler model of research utilization to facilitate evidence-based practice. Nursing Outlook, 49(6), 272–279. https://doi.org/10.1067/mno.2001.120517
Tandon, R., Lenderking, W. R., Weiss, C., Shalhoub, H., Barbosa, C., Chen, J., Greene, M., Meehan, S. R., Duvold, L., Arango, C., Agid, O., & Castle, D. (2020). The impact on functioning of second-generation antipsychotic medication side effects for patients with schizophrenia: A worldwide, cross-sectional, web-based survey. Annals of General Psychiatry, 19(1). https://doi.org/10.1186/s12991-020-00292-5
Appendix A

Appendix B
Appendix C

Appendix D
Pre-test
Multiple Choice (Select the most appropriate answer)
1. Years in clinical practice as a psychiatric provider:
☐ <1 year ☐ 1–5 years ☐ 6–10 years ☐ >10 years
2. Primary patient population served (select all that apply):
☐ Adults ☐ Adolescents ☐ Children
- Approximate number of patients prescribed SGAs per week:
☐ 1–5 ☐ 6–10 ☐ >10
Response scale: Never / Rarely / Sometimes / Often / Always
- I routinely obtain baseline metabolic measurements when initiating SGAs.
- I routinely monitor weight and BMI in patients prescribed SGAs.
- I routinely assess blood pressure in patients receiving SGAs.
- I routinely review or order fasting glucose and lipid panels when indicated.
8. What barriers limit routine metabolic monitoring in your practice? (Select all that apply)
☐ Time constraints
☐ Access to laboratory testing
☐ Unclear role between psychiatry and primary care
☐ Patient nonadherence
☐ Lack of standardized tools
☐ Limited confidence or knowledge
☐ Other: ___________
9. Second-generation antipsychotics (SGAs) are most associated with which adverse condition?
a. Cancer
b. Metabolic Syndrome
c. Addison’s Disease
d. Parkinson’s Disease
10. According to ADA/APA guidelines, which parameters should be included in baseline and follow-up metabolic screening for patients on SGAs?
a. Weight, fasting glucose, and blood pressure only
b. Waist circumference, blood pressure, fasting glucose, and lipids
c. Liver function tests and EKG
d. CBC and urinalysis
11. Which SGAs are associated with the highest risk of metabolic side effects such as weight gain and insulin resistance?
a. Aripiprazole and Risperidone
b. Clozapine and Olanzapine
c. Ziprasidone and Lurasidone
d. Haloperidol and Fluphenazine
12. According to the literature, what is the approximate range of MetS prevalence in patients on SGAs?
a. 5–10%
b. 10–25%
c. 32–68%
d. 70–90%
13. Which blood pressure reading meets the threshold for metabolic syndrome?
a. 115/70
b. 125/82
c. 130/85
d. 120/75
14. Which waist circumference measurement indicates increased metabolic risk in women?
a. 30 inches
b. 35 inches
c. 40 inches
d. 45 inches
15. Which fasting glucose value is considered diagnostic for Metabolic Syndrome?
a. 89 mg/dL
b. 99 mg/dL
c. 102 mg/dL
d. 95 mg/dL
16. Best practice recommends reassessing weight after SGA initiation at:
a. Every 6 months
b. At 4, 8, and 12 weeks
c. Only at baseline and annually
d. Monthly indefinitely
17. What aspects of the educational session were most useful?
18. What barriers do you anticipate in implementing consistent metabolic monitoring?
19. What resources or changes would help facilitate improved monitoring?
Appendix E
Post-test
Multiple Choice (Select the most appropriate answer)
8. Years in clinical practice as a psychiatric provider:
☐ <1 year ☐ 1–5 years ☐ 6–10 years ☐ >10 years
9. Primary patient population served (select all that apply):
☐ Adults ☐ Adolescents ☐ Children
- Approximate number of patients prescribed SGAs per week:
☐ 1–5 ☐ 6–10 ☐ >10
Response scale: Never / Rarely / Sometimes / Often / Always
- I routinely obtain baseline metabolic measurements when initiating SGAs.
- I routinely monitor weight and BMI in patients prescribed SGAs.
- I routinely assess blood pressure in patients receiving SGAs.
- I routinely review or order fasting glucose and lipid panels when indicated.
8. What barriers limit routine metabolic monitoring in your practice? (Select all that apply)
☐ Time constraints
☐ Access to laboratory testing
☐ Unclear role between psychiatry and primary care
☐ Patient nonadherence
☐ Lack of standardized tools
☐ Limited confidence or knowledge
☐ Other: ___________
9. Second-generation antipsychotics (SGAs) are most associated with which adverse condition?
a. Cancer
b. Metabolic Syndrome
c. Addison’s Disease
d. Parkinson’s Disease
10. According to ADA/APA guidelines, which parameters should be included in baseline and follow-up metabolic screening for patients on SGAs?
a. Weight, fasting glucose, and blood pressure only
b. Waist circumference, blood pressure, fasting glucose, and lipids
c. Liver function tests and EKG
d. CBC and urinalysis
11. Which SGAs are associated with the highest risk of metabolic side effects such as weight gain and insulin resistance?
a. Aripiprazole and Risperidone
b. Clozapine and Olanzapine
c. Ziprasidone and Lurasidone
d. Haloperidol and Fluphenazine
13. According to the literature, what is the approximate range of MetS prevalence in patients on SGAs?
a. 5–10%
b. 10–25%
c. 32–68%
d. 70–90%
14. Which blood pressure reading meets the threshold for metabolic syndrome?
a. 115/70
b. 125/82
c. 130/85
d. 120/75
15. Which waist circumference measurement indicates increased metabolic risk in women?
a. 30 inches
b. 35 inches
c. 40 inches
d. 45 inches
16. Which fasting glucose value is considered diagnostic for Metabolic Syndrome?
a. 89 mg/dL
b. 99 mg/dL
c. 102 mg/dL
d. 95 mg/dL
17. Best practice recommends reassessing weight after SGA initiation at:
a. Every 6 months
b. At 4, 8, and 12 weeks
c. Only at baseline and annually
d. Monthly indefinitely
18. What aspects of the educational session were most useful?
19. What barriers do you anticipate in implementing consistent metabolic monitoring?
20. What resources or changes would help facilitate improved monitoring?
Appendix E
1 month follow-up
Multiple Choice (Select the most appropriate answer)
1. Years in clinical practice as a psychiatric provider:
☐ <1 year ☐ 1–5 years ☐ 6–10 years ☐ >10 years
2. Primary patient population served (select all that apply):
☐ Adults ☐ Adolescents ☐ Children
- Approximate number of patients prescribed SGAs per week:
☐ 1–5 ☐ 6–10 ☐ >10
Response scale: Never / Rarely / Sometimes / Often / Always
- I routinely obtain baseline metabolic measurements when initiating SGAs.
- I routinely monitor weight and BMI in patients prescribed SGAs.
- I routinely assess blood pressure in patients receiving SGAs.
- I routinely review or order fasting glucose and lipid panels when indicated.
8. What barriers limit routine metabolic monitoring in your practice? (Select all that apply)
☐ Time constraints
☐ Access to laboratory testing
☐ Unclear role between psychiatry and primary care
☐ Patient nonadherence
☐ Lack of standardized tools
☐ Limited confidence or knowledge
☐ Other: ___________
9. Second-generation antipsychotics (SGAs) are most associated with which adverse condition?
a. Cancer
b. Metabolic Syndrome
c. Addison’s Disease
d. Parkinson’s Disease
10. According to ADA/APA guidelines, which parameters should be included in baseline and follow-up metabolic screening for patients on SGAs?
a. Weight, fasting glucose, and blood pressure only
b. Waist circumference, blood pressure, fasting glucose, and lipids
c. Liver function tests and EKG
d. CBC and urinalysis
11. Which SGAs are associated with the highest risk of metabolic side effects such as weight gain and insulin resistance?
a. Aripiprazole and Risperidone
b. Clozapine and Olanzapine
c. Ziprasidone and Lurasidone
d. Haloperidol and Fluphenazine
13. According to the literature, what is the approximate range of MetS prevalence in patients on SGAs?
a. 5–10%
b. 10–25%
c. 32–68%
d. 70–90%
14. Which blood pressure reading meets the threshold for metabolic syndrome?
a. 115/70
b. 125/82
c. 130/85
d. 120/75
15. Which waist circumference measurement indicates increased metabolic risk in women?
a. 30 inches
b. 35 inches
c. 40 inches
d. 45 inches
16. Which fasting glucose value is considered diagnostic for Metabolic Syndrome?
a. 89 mg/dL
b. 99 mg/dL
c. 102 mg/dL
d. 95 mg/dL
17. Best practice recommends reassessing weight after SGA initiation at:
a. Every 6 months
b. At 4, 8, and 12 weeks
c. Only at baseline and annually
d. Monthly indefinitely
18. What aspects of the educational session were most useful?
19. What barriers do you anticipate in implementing consistent metabolic monitoring?
20. What resources or changes would help facilitate improved monitoring?
